Why Do Some Fillings Require the Option of an Inlay and What Is the Difference Between an Inlay and a Filling?
2024-12-26
2026-02-16
3D printed dental surgical guides have become essential in North American implantology and restorative dentistry by 2026. These patient-specific templates—designed from CBCT and intraoral scans—ensure precise implant placement, reducing risks and improving outcomes. In the US and Canada, adoption grows rapidly due to digital workflow maturity, rising implant demand, and regulatory clarity from the FDA.
While 3D printing excels at complex geometries and customization, CAD file compliance and adaptation to CAM milling remain critical for hybrid workflows: printing guides/models and milling definitive zirconia restorations. This article examines compliance requirements, CAD-to-CAM adaptation, accuracy benefits, trends, and best practices for North American labs and clinics.
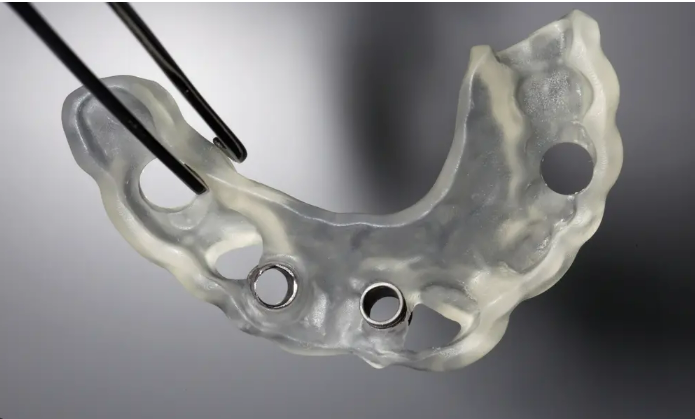
The FDA regulates 3D printed dental devices—including surgical guides—as medical devices under the Center for Devices and Radiological Health (CDRH). Guides are typically Class II (moderate risk), requiring 510(k) clearance for commercial distribution or point-of-care manufacturing under specific conditions.
Key Compliance Requirements (2026):
Non-compliance risks enforcement actions; many clinics partner with cleared manufacturers or use FDA-cleared resins/systems.
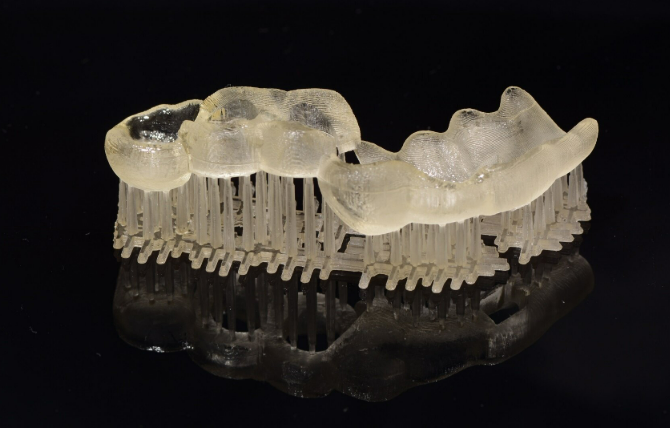
Many North American labs print guides/models and mill zirconia finals. Adaptation ensures seamless transition.
Key Adaptation Steps:
Benefits: Hybrid reduces waste (additive for complex guides, subtractive for strength); improves fit via guided placement.

In 2026 North America, 3D printed dental guides require strict CAD file compliance (FDA validation) and smart CAM milling adaptation for hybrid success. Clinics and labs mastering these achieve precise implant placement, durable zirconia restorations, and regulatory safety—driving efficiency and patient outcomes across the US and Canada.

Dry & wet milling for zirconia, PMMA, wax with auto tool changer.
learn more
High-precision 3D scanning, AI calibration, full-arch accuracy.
learn more
40-min full sintering with 57% incisal translucency and 1050 MPa strength.
learn more

40-min cycle for 60 crowns, dual-layer crucible and 200°C/min heating.
learn more
High-speed LCD printer for guides, temporaries, models with 8K resolution.
learn more